Regulatory Science & Product Development Experts helping customers
Commercialise Products | Reduce Time to Market | Achieve Compliance | Grow Business
New Drugs
Vyomus Consulting is a regulatory and development partner helping pharmaceutical and biopharmaceutical companies commercialize New Drugs, Investigational New Drugs and Subsequent New Drugs be it APIs or finished formulations in India. From discovery to commercialization, we design and implement efficient and effective regulatory and product development solutions which integrate science, regulations and business objectives, to overcome product commercialisation challenges.
Any regulated medicinal product which is to be imported, manufactured, stored, distributed, tested or sold in India, has to be registered with the licensing authority, Central Drugs Standard Control Organization (CDSCO) or specific State Licensing Authorities through various procedures.
CDSCO designates any pharmaceutical product as a New Drug, Investigational New Drugor Subsequent New drug based on the following parameters :
- A bulk drug substance, finished product which has not been used in the country to any significant extent and has not been recognized as safe and effective.
- A drug already approved by CDSCO for certain claims, which is now proposed to be marketed with modified or new claims, indications, dosage, dosage form (including sustained release dosage form) and route of administration.
This new drug designation shall continue to be considered as new drug for a period of four years from the date of its first approval. The procedures and processes for registering New Drug, Investigational New Drug or Subsequent New drug products are clearly defined and outlined under appropriate regulations.
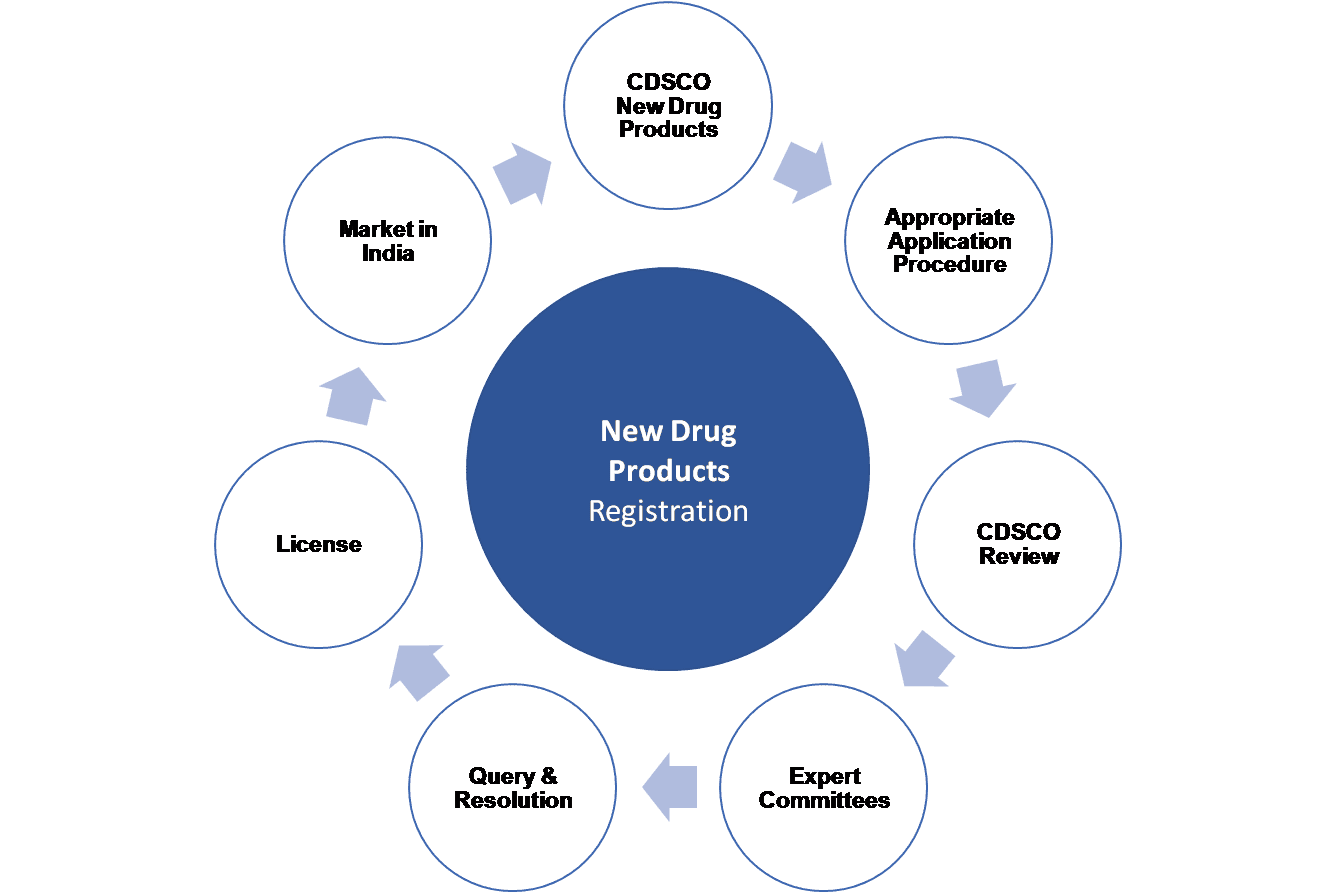
Vyomus Consulting provides end-to-end Product Registration Services as well as Authorised Agent Services to all importers and manufactures of New Drugs, Investigational New Drugs or Subsequent New Drugs. Our service offerings will help you import, manufacture, test, conduct clinical trials and distribute your products in India (Market Authorisation), efficiently so as to achieve faster product approvals leading to enhanced access to Markets and Opportunities.
Our senior practitioners have a unique blend of business, science, regulations and technology expertise and include experts in product development, regulatory affairs, pharmacology, toxicology, preclinical and clinical trial design and evaluation, biostatistics, each with extensive product commercialisation experience.
Regulatory Consulting, Regulatory Strategy, Preclinical Study Approvals, Clinical Study Approvals, Market Authorisations, Labelling Services, Regulatory Submissions, Regulatory Reporting, Regulatory Intelligence, Authorised Agent Services.

