Regulatory Science & Product Development Experts helping customers
Commercialise Products | Reduce Time to Market | Achieve Compliance | Grow Business
Regulatory Intelligence
In general, regulatory intelligence can bedefined as the monitoring, gathering and analysing of publicly available and experience based regulatory information including communicating the implications of that information, and monitoring the current regulatory environment for opportunities to shape future regulations, guidance, policy, legislationandhence develop a strategy for time and costefficient drug development.
Minimally, regulatory intelligence should be responsible for impact assessments, stakeholder identification, prioritization, regulatory trending, follow-up, and evaluation.
Vyomus Consulting regulatory practitioners analyse your specific request, create a product specific regulatory intelligence process document, help customers fully understand the regulatory environment and develop an advantage focused optimised strategy for : Feasibility of clinical studies, Adaption of development program, Answering regulatory & product development questions, Regulatory requirement overview, Preparation for regulatory meetings, Corporate Education/training, Targeted alerts etc.,
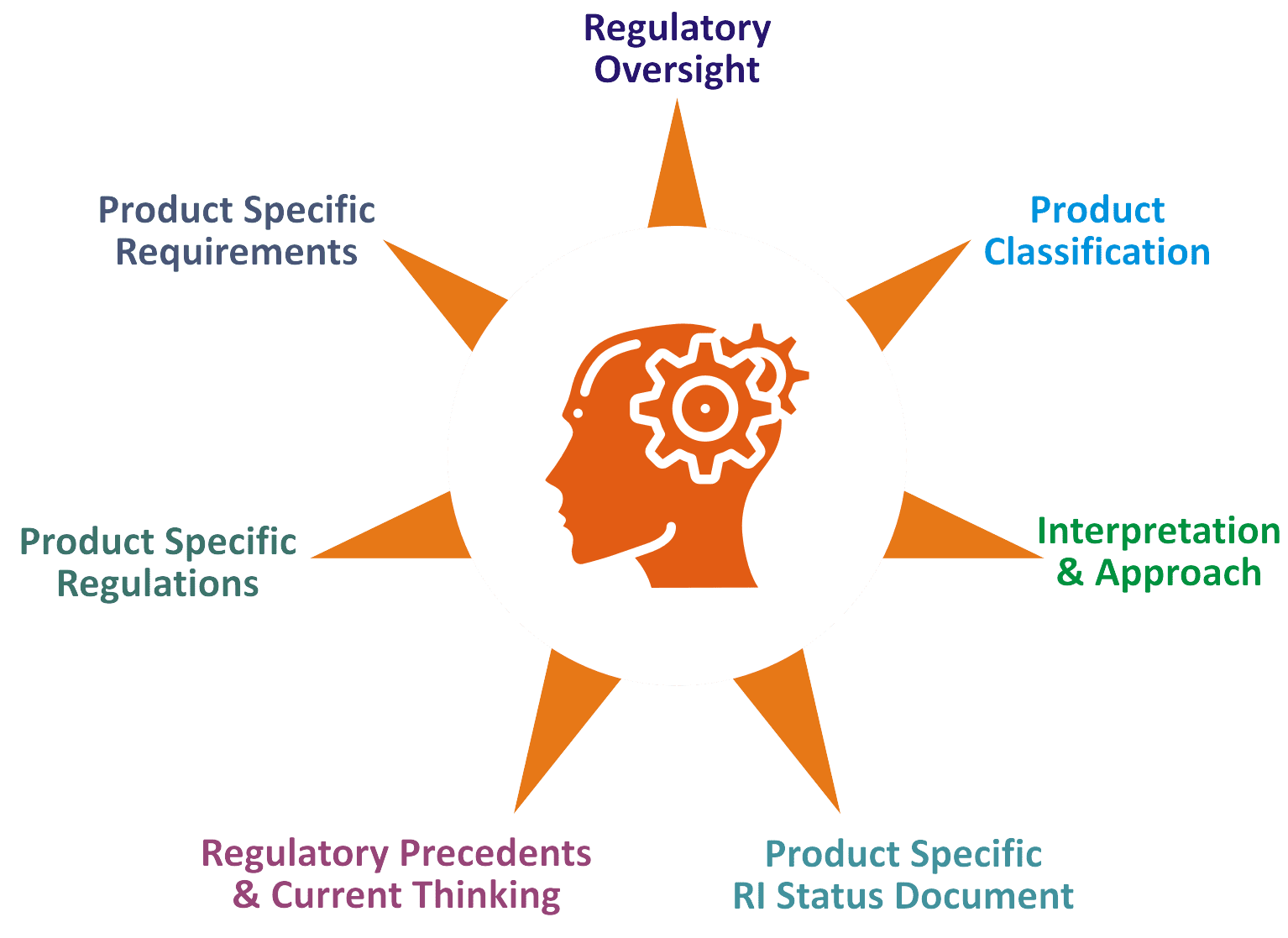
Integrating Science, Regulations, Measurable Business outcomes and combining local knowledge with global standards, we move beyond providing expert advice to get your project off the ground, move at a fast pace and achieve faster approvals.
Our overall experience includes over six hundred preclinical, clinical trial approvals, product registrations, marketing authorizations, import and marketing authorizations, manufacturing and marketing authorization and transactions covering a large number of therapeutic and product categories.
Industry focus :Pharmaceuticals, Medical Devices, Cosmetics & Wellnes, Food & Nutrition.
- Act as your Regulatory Intelligence Department,
- Provide precedence research and analysis for a therapeutic indication,
- Conduct research and/or global surveillance,
- Set up your RI department and/or manage RI information for your company,
- Write and publish newsletters specific to your company’s needs,
- Conduct comparative RI across the requirements for specific countries.

