Regulatory Science & Product Development Experts helping customers
Commercialise Products | Reduce Time to Market | Achieve Compliance | Grow Business
Cosmetics
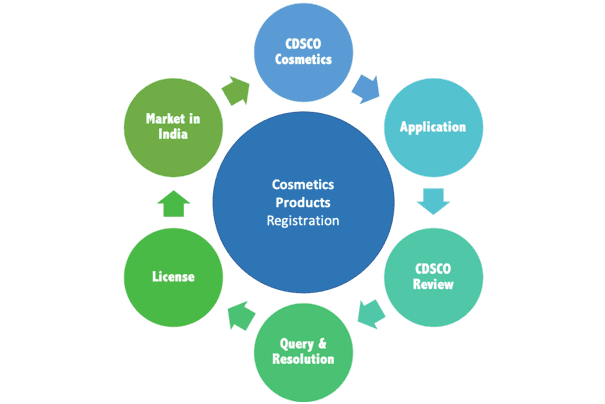
In order to legally register, import and market, sell your cosmetic products in India you need to be compliant to Indian regulations and legislation. The government of India has notified the requirement for registration of “Import (for sale) of Cosmetics” through its Gazette Notification G.S.R 426(E) dated 19th May 2010. This regulation was implemented from 1st April 2013.
In summary, cosmetics manufacturer/s who wish to import and market their products in India, have to register their manufacturing site/s and product/s with the Indian Drug & Cosmetics Regulatory Agency (licensing agency) – CDSCOthrough their registered office in India, or their Subsidiary or through an Importer or an India based Authorized Agent.
Only one Indian Authorized Representative office is needed for the whole country. Vyomus Consulting will represent you legally in India so that all your distributors can buy the products from us, although activities like distributor identification, product marketing, product sales etc., are not being supported at present.
Vyomus Consulting provides end-to-end Product Registration, Authorised Agent, Product Categorisation, Ingredients Evaluation, Claims Support and Product Testing services. An end-to-end service offering which will help you import and sell your product in India, so as to achieve faster product approvals leading to enhanced access to Markets and Opportunities.